How This Baby Supply Maker Dealt with Toxic Teether
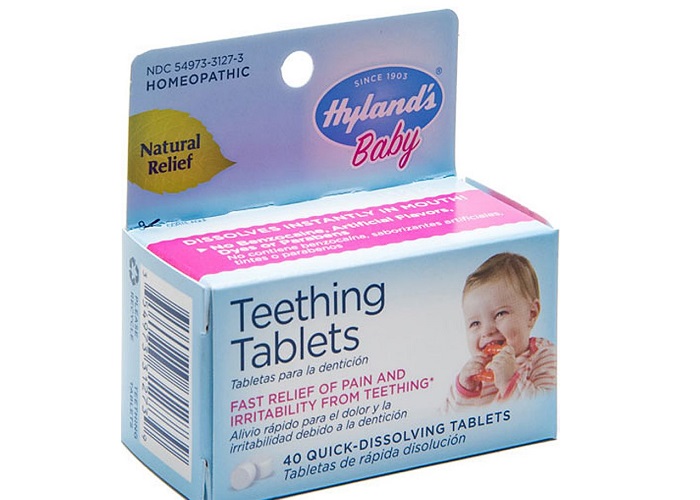
US regulators advised Highland to pull and recall its teething tablets due to a “potentially toxic” level of herbs in the medicine. Standard Homeopathic Co. of Los Angeles, the company that makes the Highland brand teething tablet quickly complied with the request, issuing a recall of an unspecified quantity of the products. They also ceased manufacturing the product last fall, and now they have notified distributors to pull any products currently on the shelves.
The product itself is a common one, used to help relieve the pain of emerging teeth in infants. Companies do have these issues, and they certainly reacted quickly, but that doesn’t mean this problem won’t hurt their public relations.
The Highland brand is already a niche product for a target market of consumers who want a less mainstream, more homeopathic option in taking care of their children. These parents are making a conscious choice to do what they believe is healthier for their babies, and they are doing it because they want to know they are doing all they can to keep their baby healthy.
To learn that the product they’re giving their child could potentially be toxic is a major blow. They are choosing Highland specifically because they believe mainstream products might be more toxic. Now they’re being told what they chose actually is. That presents a very difficult emotional scenario … and it could be tough for some self-described “crunchy moms” to get past.
The Homeopathic industry is a huge business in the United States. While many products stand up to rigorous testing, some don’t carry the same level of quality control as other products on the market. Highland , as it has been tested and these products have been eliminated, clearly is one of the products that is undergoing additional quality control. That’s certainly a mark in their favor, and it’s a message they need to send to their current and prospective customers, loud and clear: “We had an issue, and, because we are being careful, we caught it and are fixing it as best we can.”
In an effort to catch up and get ahead of the curve on regulation of this market, the FDA is working overtime to investigate products that fall under the homeopathic or “alternative health” umbrella. Back in September, the FDA issued a warning that teething tablets containing the herb, belladonna, should not be used. This herb, known as nightshade, is poisonous in certain amounts … but it has remained a mainstay in homeopathic circles for centuries.
While this situation will likely fan the flames of controversy, Highland has the opportunity here to say: “We’re on the side of being safe.” Despite the bad news that started all this, they are in the position to lead the way going forward … if they choose to accept that opportunity.
Discover more from Ronn Torossian
Ronn Torossian’s Professional Profile on Muck Rack
GuideStar Profile for Ronn Torossian Foundation
Ronn Torossian’s Articles on Entrepreneur
Ronn Torossian’s Blog Posts on Times of Israel
Ronn Torossian on SoundCloud
US regulators advised Highland to pull and recall its teething tablets due to a “potentially toxic” level of herbs in the medicine. Standard Homeopathic Co. of Los Angeles, the company that makes the Highland brand teething tablet quickly complied with the request, issuing a recall of an unspecified quantity of the products. They also ceased manufacturing the product last fall, and now they have notified distributors to pull any products currently on the shelves. The product itself is a common one, used to help relieve the pain of emerging teeth in infants. Companies do have these issues, and they certainly reacted quickly, but that doesn’t mean this problem won’t hurt their public relations. The Highland brand is already a niche…